PROTECT YOUR DNA WITH QUANTUM TECHNOLOGY
Orgo-Life the new way to the future Advertising by Adpathway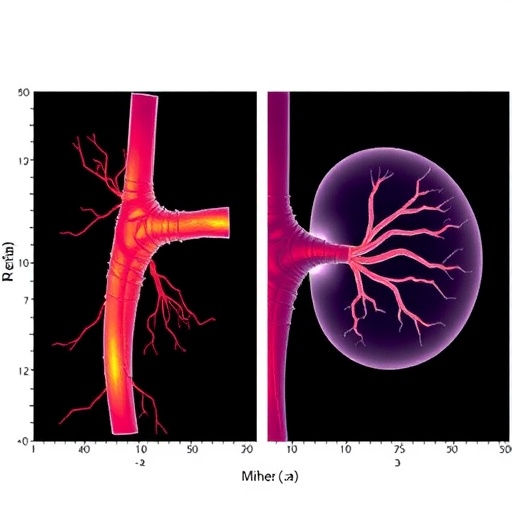
In a groundbreaking study published in the “Annals of Biomedical Engineering,” researchers led by Corvo et al. explore a novel computational simulation that reveals previously uncharted dynamics of renal arteries following endovascular aneurysm repair (EVAR). This cutting-edge research seeks to understand how respiration-induced deformations influence the complex blood flow within these vital vessels, shedding light on a topic previously underrepresented in vascular biomechanical studies. This study not only pushes the boundaries of computational modeling but also holds potential implications for improving surgical outcomes and vascular health.
The renal arteries are key players in the human circulatory system, supplying blood to the kidneys, regulating blood pressure, and maintaining electrolyte balance. Their proper function is essential for renal health, and any aberration in their biomechanics can lead to significant health complications. In the context of EVAR, a minimally invasive surgical procedure implemented to treat abdominal aortic aneurysms, understanding how these arteries deform under physiological conditions is critical. EVAR can create altered flow conditions that may lead to unintended anatomical and functional changes in adjacent vessels, particularly under the dynamic influence of breathing.
Corvo and his team set out to develop a sophisticated computational model to simulate the impact of respiratory movement on the renal arteries post-EVAR. The researchers utilized advanced finite element analysis techniques, a cornerstone of modern engineering simulations, to accurately replicate the mechanical properties and behavior of the renal arteries during respiratory cycles. This approach incorporated detailed anatomical data obtained from medical imaging, allowing for significant fidelity in the model’s recreation of actual physiological conditions.
One of the significant challenges faced by the researchers was the variability in individual anatomy and biomechanical properties of the renal arteries. The study emphasized the importance of personalized modeling, where the anatomical variations among patients could significantly influence the outcomes of EVAR procedures. By employing high-resolution imaging techniques, they were able to create personalized vascular models that reflect the unique biomechanics of different patients, adding a layer of precision to their simulation.
The results of the simulations revealed nuanced insights into the deformations of the renal arteries during the respiratory cycle. Notably, the study demonstrated that respiratory-induced movement caused significant alterations in the flow patterns within the renal arteries, which could impact renal perfusion and, subsequently, kidney function. Understanding these dynamic changes is crucial, particularly in the context of patients who have undergone EVAR, as unconventional flow dynamics can inadvertently lead to complications such as renal ischemia or graft failure.
Beyond elucidating the mechanics of blood flow, Corvo et al. explored how these simulations could guide clinical practices regarding patient monitoring post-EVAR. By predicting how the renal arteries may respond to respiratory movements, healthcare providers can better anticipate the risks associated with vascular remodeling and help tailor post-operative care plans. This predictive modeling could lead to enhanced patient safety and improved intervention strategies in vascular surgery.
The interdisciplinary nature of this research highlights a growing trend in biomedical engineering, where the integration of computer science, biology, and engineering is paving the way for smarter, more responsive medical solutions. The application of computational simulations in understanding biomechanical phenomena exemplifies the potential of digital technologies to innovate traditional medical paradigms. With continued advancements in computational power and imaging techniques, the future of personalized medicine is increasingly bright.
Moreover, corroborating the findings from this computational study with real-world clinical data remains an essential next step. As researchers continue to validate these models against empirical observations, the potential for refining surgical training and enhancing procedural outcomes becomes more tangible. The study lays a foundation for future investigations that may leverage computational simulations to address other vascular pathologies and improve therapeutic strategies across a diverse range of conditions.
In summary, Corvo et al.’s research represents an important advancement in the field of biomedical engineering and vascular surgery, bridging the gap between computational modeling and practical clinical application. As the study underscores, understanding the complex mechanics at play in renal arteries post-EVAR not only enhances our comprehension of vascular biomechanics but also arms clinicians with crucial information that could lead to better patient outcomes. This paradigm shift toward data-driven, personalized medical approaches encapsulates the essence of modern medicine, fostering innovations that can transform surgical practices and ultimately improve the quality of care for patients worldwide.
To put it succinctly, the implications of this research extend beyond just the realm of renal arterial dynamics post-EVAR; they open avenues for exploring surgical techniques with an eye towards optimizing procedures. As we gain more insights into the mechanical behavior of arteries under various conditions, the healthcare community stands on the cusp of potentially revolutionizing both patient care and surgical approaches through the intelligent application of computational simulations.
In conclusion, the study by Corvo et al. stands as a testament to what can be achieved when interdisciplinary collaboration fuels research. It invites the broader scientific community to reconsider long-held assumptions about vascular dynamics and encourages further exploration that may lead us to groundbreaking discoveries in medical science. The future of personalized medicine is here, and its trajectory promises to enhance not only surgical interventions but the entire landscape of healthcare delivery.
Subject of Research: Computational simulation of renal artery deformation due to respiration after EVAR.
Article Title: Computational Simulation of Respiration-Induced Deformation of Renal Arteries After EVAR.
Article References:
Corvo, A., Avril, S., Aliseda, A. et al. Computational Simulation of Respiration-Induced Deformation of Renal Arteries After EVAR.
Ann Biomed Eng (2025). https://doi.org/10.1007/s10439-025-03806-y
Image Credits: AI Generated
DOI: 10.1007/s10439-025-03806-y
Keywords: Computational simulation, renal arteries, EVAR, biomechanics, personalized medicine, vascular dynamics.
Tags: biomechanics of renal arteriesblood flow simulation in renal arteriescomputational modeling in vascular engineeringendovascular aneurysm repair dynamicsimplications for surgical outcomesminimally invasive surgical techniquesrenal artery biomechanicsrenal artery shape deformationrenal health and blood pressure regulationrespiratory effects on vascular healthrespiratory-induced vascular changesvascular biomechanical studies


 1 day ago
16
1 day ago
16



















 English (US) ·
English (US) ·  French (CA) ·
French (CA) ·